Allergan Receives FDA Approval for XEN Glaucoma Treatment System
On November 22, 2016, Allergan announced that the U.S. Food and Drug Administration (FDA) has cleared the XEN® Glaucoma Treatment System for use in the United States.
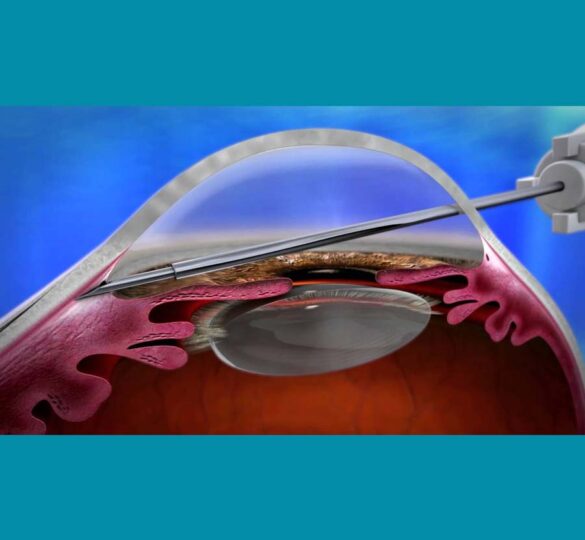
On November 22, 2016, Allergan announced that the U.S. Food and Drug Administration (FDA) has cleared the XEN® Glaucoma Treatment System (consisting of the XEN45 Gel Stent and the XEN Injector) for use in the United States.
The XEN Glaucoma Treatment System reduces intraocular pressure (IOP) in patients and is indicated for the management of refractory glaucoma, where previous surgical treatment has failed, or in patients with primary open angle glaucoma, and pseudoexfoliative or pigmentary glaucoma with open angles that are unresponsive to maximum tolerated medical therapy.
The XEN device is implanted through an ab interno approach and reduces IOP by creating a new drainage channel with a permanent implant that becomes flexible. This provides yet another new treatment option for glaucoma, and is considered a minimally invasive glaucoma surgery (MIGS) procedure.
“XEN is a new option that provides an opportunity for surgical intervention in refractory glaucoma patients,” said Robert N. Weinreb, MD, chairman and distinguished professor of Ophthalmology at the University of California, San Diego. “XEN can effectively lower IOP; in fact, studies have shown that at 12 months using XEN, patients used, on average, less IOP lowering drops than they did before XEN was implanted,” he said.
Allergan plans to launch the XEN Glaucoma Treatment System in the U.S. in early 2017. More than 10,500 XEN Gel Stents have already been distributed worldwide. XEN is CE marked in the European Union, where it is indicated for the reduction of intraocular pressure in patients with primary open angle glaucoma where previous medical treatments have failed. It is also licensed for use in Canada, Switzerland, and Turkey.
Source: Allergan